 So, when the uncertainties in momentum and position of a particle are multiplied, it is ≥ h/4pi. The uncertainty principle uses the momentum and position factor of a particle, where the momentum is equal to the mass multiplied by the velocity. Thus, the uncertainty principle is relevant only for the objects with small sizes like an atom. This is because the uncertainty, in this case, is too small. But we can easily calculate the position and velocity of a car. The Heisenberg uncertainty principle actually said that for any object in the universe, both the exact position and the exact velocity can not be calculated at the same time even in theory. So, the orbitals actually show the shape of the region where an electron can be found 95% of the time. From this, we can say that it is not possible to calculate the position and momentum of an electron with absolute precision. 
The Heisenberg Uncertainty Principle says that it is not possible to find the current position of an electron and its position an instant later. The s orbital has the lowest energy as it is closest to the nucleus while the d orbital has the maximum energy as it is farthest from the nucleus. They are the s orbital, p orbital, d orbital, and f orbital. 
Their movement is subjected to quantum mechanics, and it is described using 3-dimensional clouds known as orbitals.Ī total of four different orbitals have been discovered to date. Unlike the fixed-orbit path of planets around the sun, electrons present in an atom don't follow a definite path. 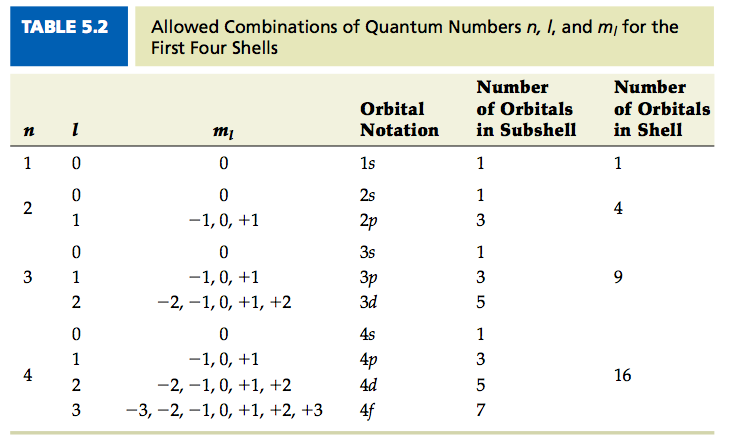
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
